In a recent article, I discussed the astounding role of motor proteins in eukaryotic cell division. But this is just one of many incredible engineered features associated with mitosis. In this and a subsequent article, I will provide an overview of the elegant molecular mechanisms that underlie the spindle assembly checkpoint and discuss the implications of its dysfunction.
Without this exquisitely engineered system, the cell risks distributing an uneven number of chromosomes to the daughter cells, potentially resulting in cancer or (in the case of meiosis), trisomy conditions such as Down Syndrome (which is characterized by an extra copy of chromosome 21).
What Is Mitosis?
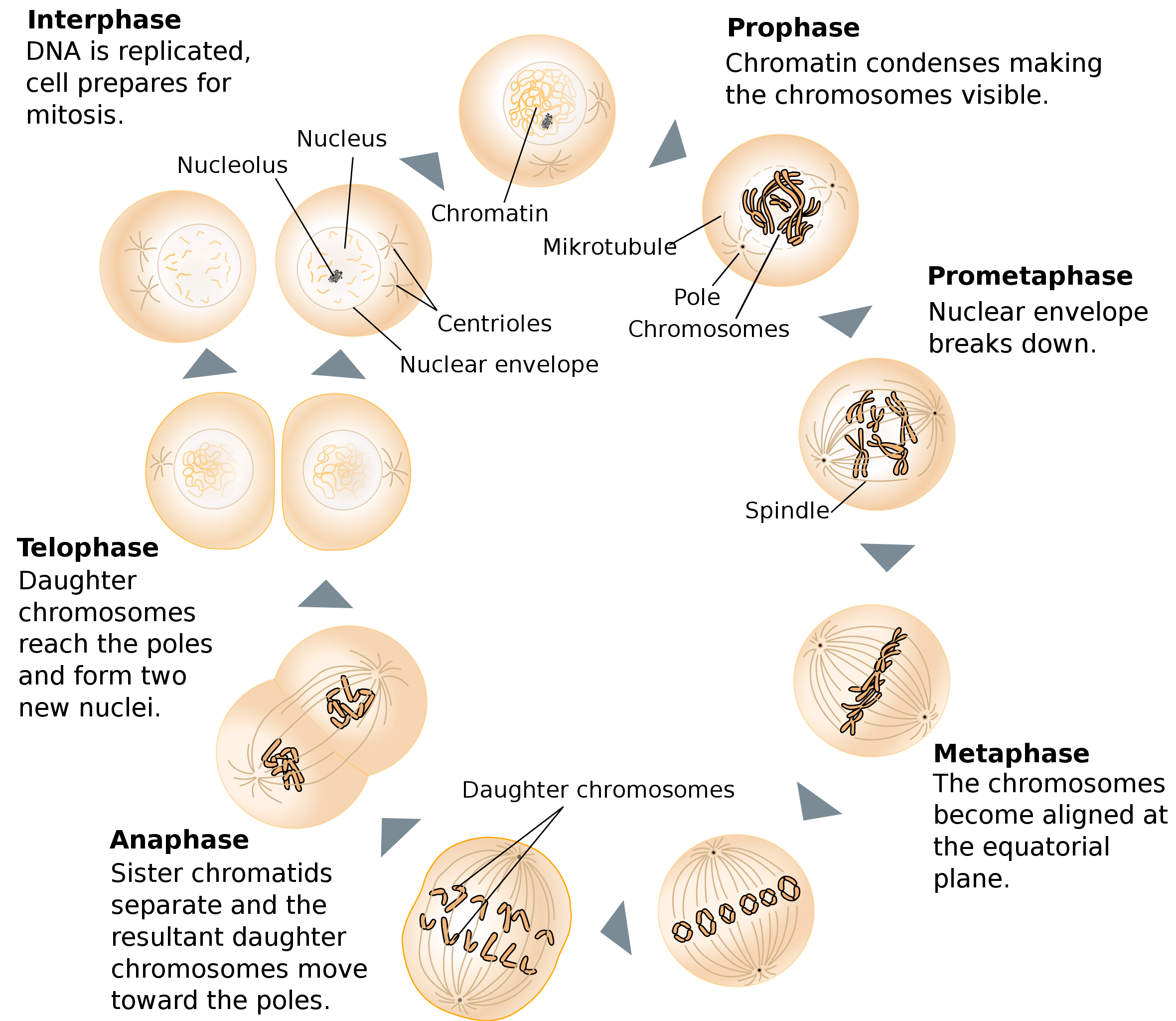
Image credit: Wikimedia Commons.
Mitotic division (“M phase”) is the culmination of the eukaryotic cell cycle for somatic cells. Mitotic cell division is divided into six phases, illustrated in the figure above. The first is prophase, which is characterized by chromosome condensation (the reorganization of the sister chromatids into compact rod-like structures). Following condensation, assembly of the mitotic spindle apparatus occurs outside the nucleus between the two centrosomes which have duplicated and moved apart to the poles of the cell.
The second stage of mitosis is prometaphase, which is marked by the disintegration of the nuclear envelope. This is followed by metaphase, where sister chromatids are attached to opposite spindle poles by microtubules bound to protein complexes called kinetochores. In animal cells, 10-40 microtubule-binding sites are associated with any one kinetochore. In yeast, each kinetochore contains only one attachment site. At this point, the chromosomes are seen to be aligned at the cell’s equator (the metaphase plate). The sister chromatids are themselves held together by the protein cohesin.
At anaphase, the sister chromatids separate to form two daughter chromosomes that are pulled towards opposite poles of the spindle. Microtubules bound to kinetochores, as well as the centrosome, are reeled in towards the cell’s periphery by specialized dynein motor proteins that “walk” towards the minus end of the microtubule but are held stationary by cargo-binding domains that are anchored to the cell cortex.
The next phase in the cycle is telophase, the stage at which the daughter chromosomes de-condense at the spindle poles and a new nuclear envelope is assembled. A contractile ring is then formed, marking the final stage of the process — cytokinesis. The contractile ring is comprised of actin and myosin filaments. The cell thus differentiates to form two new daughter cells, each with a nucleus containing a complete and identical set of chromosomes.
The consequences of improper attachment can be catastrophic, with segregation of two chromosome copies to a single daughter cell. The spindle assembly checkpoint pathway is responsible for inhibiting progression of mitosis from metaphase to anaphase until each of the sister chromatids has become correctly bi-oriented and securely associated with the mitotic spindle.
Controlling Metaphase-to-Anaphase Progression
Progression from metaphase to anaphase is mediated by the anaphase promoting complex or cyclosome (APC/C), an E3 ubiquitin ligase. When bound to a protein, Cdc20, the APC functions to ubiquitinate securin (a protein that prevents the cleavage of cohesin by the enzyme separase), as well as the S and M cyclins, thereby targeting them for destruction. [1,2,3] The APC/C is phosphorylated by cyclin dependent kinases (Cdks), thus rendering it able to bind to Cdc20 and form the APCCdc20 complex. The APCCdc20 complex is autoinhibitory, since destruction of Cdks results in a decreased rate of APC/C phosphorylation and, as a consequence, binding of Cdc20.
Microtubule attachment to kinetochores during prometaphase is governed by a “search and capture” mechanism. [4,5,6] The property of dynamic instability facilitates the process by which microtubules “search” for kinetochore attachment sites. When a microtubule encounters a kinetochore, the kinetochore is “captured” by means of side-on attachment. The sister chromatids are subsequently positioned at one of the poles of the cell, where more microtubules become attached. After the kinetochore becomes associated with a microtubule from the other pole, the chromosomes move to the equator. Though this process has been viewed for decades as being stochastic, recent work has suggested that it may in fact be more deterministic than previously recognized (see this article for a good discussion). [7]
This checkpoint pathway relies on a specialized mechanism for monitoring the security of kinetochore-microtubule attachment. [8,9] In the case of improper attachment, the kinetochore sends out a signal — the wait anaphase signal — that inhibits activation of APCCdc20, thereby arresting metaphase-to-anaphase progression.
Monitoring Spindle-Kinetochore Attachment
The precise mechanism by which the spindle checkpoint system detects improper chromatid biorientation has not been fully elucidated. Two main hypotheses have been proposed, each with its own supporting data. [10] One proposal suggests that the system monitors the level of tension at the kinetochore. [11,12,13] Another hypothesis is that the system detects attachment of the ends of the microtubules to the kinetochore. [14] The spindle assembly checkpoint pathway most likely uses a combination of those two mechanisms.
The importance of tension sensing in the spindle assembly checkpoint was first examined in insect spermatocytes, using a micromanipulation needle to apply tension to an improperly associated chromosome. Tension resulted in the commencement of anaphase in 56 minutes, whereas it was delayed by 5 to 6 hours in the absence of tension. [15]
Aurora kinase B plays a crucial role in tension sensing, and its inhibition results in an accumulation of improperly attached kinetochores. [16,17,18,19,20] Aurora kinase B is believed to induce the inhibitory signal that destabilizes kinetochore-microtubule attachments by phosphorylating components of the kinetochore’s microtubule attachment site, including the mammalian histone-H3 variant centromere protein A (CENP-A) at serine 7. [21,22] Aurora kinase B is itself recruited to the centromere by phosphorylation of CENP-A at the same residue by Aurora kinase A. [23] When the function of Aurora kinase B is inhibited, one also observes a decrease in concentration of checkpoint components BubR1, Mad2 and CENP-E, and also an inability of BubR1 to rebind to the kinetochore following a decrease in tension at the centromere. [24] Aurora kinase B is inactivated only after correct biorientation has occurred.
The role of microtubule attachment is demonstrated by the activity of checkpoint proteins at the kinetochore. For instance, Mad2 is present on unattached kinetochores during prometaphase, but is removed from the kinetochores as they become associated with the spindle. [25] Moreover, when mammalian cells are treated with low concentrations of taxol and other microtubule-targeting drugs (thereby removing tension but retaining microtubule-kinetochore attachment), the onset of anaphase is significantly delayed. [26,27]
A Factory Assembly Line
Eukaryotic cell division is, in many respects, like a factory assembly line, complete with quality-control check points and robotic machines. The sheer number of things that need to go just right for successful division to take place without major complication renders it implausible that such an elegant process could have been produced by a gradual, unguided process.
By what mechanism is the wait anaphase signal generated? Moreover, how is the spindle assembly checkpoint turned off when proper kinetochore-microtubule attachment has been established? My next article will be taken up with these questions.
Notes
1. Zachariae, W., Nasmyth, K. (1999) Whose end is destruction: cell division and the anaphase-promoting complex. Genes and Development 13, 2039-2058.
2. Barford, D. (2011) Structural insights into anaphase-promoting complex function and mechanism. Philosophical Transactions of the Royal Society B. 366, 3605–3624.
3. Schrock MS, Stromberg BR, Scarberry L, Summers MK. APC/C ubiquitin ligase: Functions and mechanisms in tumorigenesis. Semin Cancer Biol. 2020 Dec;67(Pt 2):80-91.
4. Kirschner, M., Mitchison, T. (1986) Beyond self-assembly: From microtubules to morphogenesis. Cell 3(9), 329-342.
5. Biggins S., Murray A.W. (2001) The budding yeast protein kinase Ipl1/ Aurora allows the absence of tension to activate the spindle checkpoint. Genes and Development 15: 3118–3129.
6. Hauf, S., Watanabe, Y. (2004) Kinetochore orientation in mitosis and meiosis. Cell 119, 317-327.
7. Soares-de-Oliveira J, Maiato H. Mitosis: Kinetochores determined against random search-and-capture. Curr Biol. 2022 Mar 14;32(5):R231-R234.
8. Lara-Gonzalez, P., Westhorpe, F.G., Taylor, S.S. (2012) The Spindle Assembly Checkpoint. Current Biology 22, 966-980.
9. McAinsh AD, Kops GJPL. Principles and dynamics of spindle assembly checkpoint signalling. Nat Rev Mol Cell Biol. 2023 Aug;24(8):543-559.
10. Pinsky, B.A., Biggins, S. (2005) The spindle checkpoint: tension versus attachment. Trends in Cell Biology 15(9), 486-493.
11. Li, X., Nicklas, B. (1995) Mitotic forces control a cell-cycle checkpoint. Nature 373, 630-632.
12. Nicklas, R.B., Ward, S.C., Gorbsky, G.J. (1995) Kinetochore Chemistry Is Sensitive to Tension and May Link Mitotic Forces to a Cell Cycle Checkpoint. The Journal of Cell Biology 130(4), 929-939.
13. Larson JD, Asbury CL. Relax, Kinetochores Are Exquisitely Sensitive to Tension. Dev Cell. 2019 Apr 8;49(1):5-7.
14. Waters, J.C., Chen, R., Murray, A.W., Salmon, E.D. (1998) Localization of Mad2 to Kinetochores Depends on Microtubule Attachment, Not Tension. The Journal of Cell Biology 141, 1181-1191.
15. Li, X., Nicklas, B. (1995) Mitotic forces control a cell-cycle checkpoint. Nature 373, 630-632.
16. Adams, R.R., Maiato, H., Earnshaw, W.C., Carmena, M. (2001) Essential roles of Drosophila inner centromere protein (INCENP) and Aurora-B in histone H3 phosphorylation, metaphase chromosome alignment, kinetochore disjunction, and chromosome segregation. Journal of Cell Biology 153, 865-880.
17. Biggins S., Murray A.W. (2001) The budding yeast protein kinase Ipl1/ Aurora allows the absence of tension to activate the spindle checkpoint. Genes and Development 15: 3118–3129.
18. Kallio, M.J., McCleland, M.L., Stukenberg, P.T., Gorbsky, G.J. (2002) Inhibition of aurora B kinase blocks chromosome segregation, overrides the spindle checkpoint, and perturbs microtubule dynamics in mitosis. Current Biology 12, 900-905.
19. Tanaka T.U, Rachidi N., Janke C., Pereira G., Galova M., Schiebel E., Stark M.J., Nasmyth K. (2002) Evidence that the Ipl1-Sli15 (Aurora kinase-INCENP) complex promotes chromosome bi-orientation by altering kinetochore-spindle pole connections. Cell 108: 317–329.
20. Hauf, S., Cole, R.W., LaTerra, S., Zimmer, C., Schnapp, G., Walter, R., Heckel, A., van Meel, J., Rieder, C.L., Peters, J.M. (2003) The small molecule Hesperadin reveals a role for Aurora B in correcting kinetochore-microtubule attachment and in maintaining the spindle assembly checkpoint. Journal of Cell Biology 161, 281-294.
21. Zeitlin, S.G., Shelby, R.D., Sullivan, K.F. (2001) CENP-A is phosphorylated by Aurora B kinase and plays an unexpected role in completion of cytokinesis. Journal of Cell Biology 155, 1147-1157.
22. Liu, D., Lampson, M. (2009) Regulation of kinetochore–microtubule attachments by Aurora B kinase. Biochemical Society Transactions 37(5), 976-980.
23. Kunitoku, N., Sasayama, T., Marumoto, T., Zhang, D., Honda, S., Kobayashi, O., Hatakeyama, K., Ushio, Y., Saya, H., Hirota, T. (2003) CENP-A phosphorylation by Aurora-A in prophase is required for enrichment of Aurora-B at inner centromeres and for kinetochore function. Developmental Cell 5, 853-864.
24. Ditchfield, C., Johnson, V.L., Tighe, A., Ellston, R., Haworth, C., Johnson, T., Mortlock, A., Keen, N., Taylor, S.S. (2003) Aurora B couples chromosome alignment with anaphase by targeting BubR1, Mad2, and Cenp-E to kinetochores. Journal of Cell Biology161(2):267-80.
25. Waters, J.C., Chen, R., Murray, A.W., Salmon, E.D. (1998) Localization of Mad2 to Kinetochores Depends on Microtubule Attachment, Not Tension. The Journal of Cell Biology 141, 1181-1191.
26. Waters, J.C., Chen, R., Murray, A.W., Salmon, E.D. (1998) Localization of Mad2 to Kinetochores Depends on Microtubule Attachment, Not Tension. The Journal of Cell Biology 141, 1181-1191.
27. Hoffman, D.B., Pearson, C.G., Yen, T.J., Howell, B.J., Salmon, E.D. (2001) Microtubule-dependent changes in assembly of microtubule motor proteins and mitotic spindle checkpoint proteins at PtK1 kinetochores. Molecular Biology of the Cell 12(7), 1995-2009.
This article was originally published at Evolution News & Science Today, on August 26, 2024.